 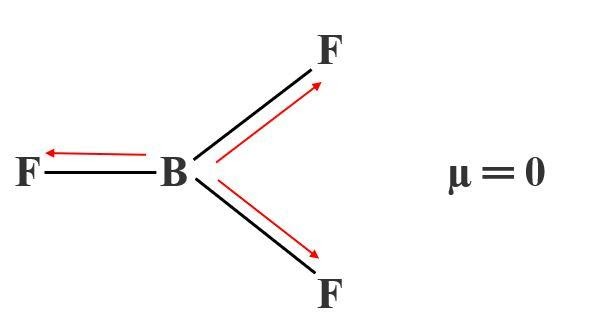
The adduct with diethyl ether, boron trifluoride diethyl etherate, or just boron trifluoride etherate, (BF 3 Tetrafluoroborate salts are commonly employed as non-coordinating anions. They undergo rapid halide exchange reactions:īecause of the facility of this exchange process, the mixed halides cannot be obtained in pure form.īoron trifluoride is a versatile Lewis acid that forms adducts with such Lewis bases as fluoride and ethers:ĬsF + BF 3 → CsBF 4 O(C 2H 5) 2 + BF 3 → BF 3 Unlike the aluminium and gallium trihalides, the boron trihalides are all monomeric. The grease used in the equipment should be fluorocarbon based, as boron trifluoride reacts with the hydrocarbon-based ones. Polytetrafluoroethylene, polychlorotrifluoroethylene, polyvinylidene fluoride, and polypropylene show satisfactory resistance. In presence of moisture it corrodes steel, including stainless steel. Suitable metals for equipment handling boron trifluoride include stainless steel, monel, and hastelloy. Storage or transport vessels should be designed to withstand internal pressure, since a refrigeration system failure could cause pressures to rise to the critical pressure of 49.85 bar (4.985 MPa). A well documented route involves the thermal decomposition of diazonium salts of BF − 4: PhN 2BF 4 → PhF + BF 3 + N 2Īlternatively it arises from the reaction of sodium tetrafluoroborate, boron trioxide, and sulfuric acid: 6 NaBF 4 + B 2O 3 + 6 H 2SO 4 → 8 BF 3 + 6 NaHSO 4 + 3 H 2O Properties Īnhydrous boron trifluoride has a boiling point of −100.3 ☌ and a critical temperature of −12.3 ☌, so that it can be stored as a refrigerated liquid only between those temperatures. Laboratory routes to the solvent-free materials are numerous. Laboratory scale įor laboratory scale reactions, BF 3 is usually produced in situ using boron trifluoride etherate, which is a commercially available liquid. Approximately 2300-4500 tonnes of boron trifluoride are produced every year. Typically the HF is produced in situ from sulfuric acid and fluorite (CaF 2). īF 3 is manufactured by the reaction of boron oxides with hydrogen fluoride: Others point to the ionic nature of the bonds in BF 3. A facile explanation invokes the symmetry-allowed overlap of a p orbital on the boron atom with the in-phase combination of the three similarly oriented p orbitals on fluorine atoms. In the boron trihalides, BX 3, the length of the B–X bonds (1.30 Å) is shorter than would be expected for single bonds, and this shortness may indicate stronger B–X π-bonding in the fluoride. The molecule is isoelectronic with the carbonate anion, CO 2− 3.īF 3 is commonly referred to as " electron deficient," a description that is reinforced by its exothermic reactivity toward Lewis bases. The molecule has no dipole moment by virtue of its high symmetry. Its D 3h symmetry conforms with the prediction of VSEPR theory. The geometry of a molecule of BF 3 is trigonal planar. It is a useful Lewis acid and a versatile building block for other boron compounds. This pungent, colourless, and toxic gas forms white fumes in moist air. Boron trifluoride is the inorganic compound with the formula BF 3. Other elements that may at times form incomplete octets include Beryllium and Aluminum.īF3 Ball and Stick Model. BF3 for example will try to form an octet on the central atom by converting to BF4. However, when these kinds of elements are handled in real life they are often unstable due to the incomplete octet. Therefore, it is simply assumed that the structure with the incomplete octet is a more stable configuration. Since double-bonding one of the fluorine's to complete the octet would leave the fluorine with a formal charge of +1, it becomes quickly apparent the infeasibility of this structure since fluorine is the most electronegative element and would not simply lose an electron. These are generally called incomplete octets and are formed due to the low electronegativity of Boron. You may have observed that this structure for BF3 seems to violate the octate rule specifically for the central Boron atom which only has six electrons in its valence shell. This results in an overall structure that is generally nonpolar in character. Answer: BF3 is nonpolar because all of the fluorine atoms are arranged in a trigonal planar fashion around the molecule, thereby cancelling out each other's pull for electrons.Īlthough the difference between Boron's electronegativity (2.04) and Fluorine's (3.98) is great enough to make the individual bonds polar covalent, the symmetrical arrangement of atoms around the central Boron atom cancels out these possible dipole moments.
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
